
The Luftwaffe was introducing 100-octane fuel at the same time, and without it the RAF could have been at a real disadvantage.Īnd Russell Marker you ask? Well, he became one of the few university professors not to have a PhD. 100 octane fuel from the refineries in Aruba, off the Venezuelan coast, found its way into squadron use with the RAF’s Hurricanes and Spitfires in 1939-40, in time for the Battle of Britain. The Supermarine Spitfire had its origins in the seaplanes that won the high-speed Schneider trophy for Britain three times in 1927-1931, when Air Commodore Rod Banks and others developed high-octane fuels for this. It may not make much difference to a car, but seventy-five years ago, it made a significant difference to aircraft engines. Another way is by adding benzene and other aromatic hydrocarbons, but benzene in particular is toxic, creating another problem. And after the fuel had been burned, toxic lead entered the environment. This was done from the 1920s onwards, but had the disadvantage of being toxic – many workers at companies making TEL died in the first decade of production. There are other ways of increasing octane ratings. They also produce branched chain alkanes, like isooctane, by isomerisation reactions. Another way of getting valuable fuels is by processes known as reforming and cracking, the latter doing the same sort of thing that happens when crude oil is formed in the first place, using heat (and catalysts) to break long chain molecules with high boiling points into smaller, more volatile, molecules which are more valuable fuels. Partly this means distilling it, and collecting the fractions with different boiling points. Having struck oil, an oil company has to refine their crude product, as it is no use as a fuel. 
The Luftwaffe was introducing 100-octane fuel at the same time, and without it the RAF could have been at a real disadvantage This mixture is crude oil, and impregnates the rock, ‘source rock’ as geologists term it. This caused the large molecules to split into a variety of smaller molecules with shorter chains. As time passed, it got buried deeper and deeper, so that eventually as the layers hardened and got turned into shale or mudstone, the organic molecules were subjected to increasing pressure and temperature. Many millions of years ago, organic matter like plankton built up on the sea floor. Now of course you can’t get molecules like octane and isooctane just by tapping them off from a tree like you can get latex to make rubber. The higher the octane rating, the more the gas/air mixture can be compressed before ignition, and the more power you will get. A fuel described as 95 octane has the same knocking characteristics as a mixture containing 95% isooctane and 5% heptane. So scientists made up mixtures with varying amounts of heptane and isooctane in order to study their knocking characteristics. Yet when he tested ordinary octane, which had a straight chain of eight carbons, that was bad too. This had a branched chain of eight carbon atoms, but the great thing was that it did not cause any knocking. Next he chose what we call 2,2,4-trimethylpentane, which at that time was usually known as isooctane. Anyway, heptane caused really bad knocking. At best they have a zig-zag chain of carbon atoms, with attached hydrogens, though at room temperature the chains will be flexing around. Chemists tend to refer to such chains as ‘straight chains’ but in fact they are anything but straight. He started by looking at heptane, a ‘straight chain’ hydrocarbon. 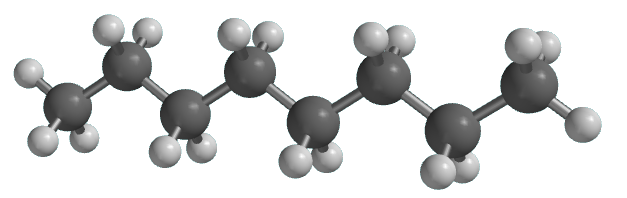
In the mid-1920s an American chemist named Russell Marker, working for the Ethyl Corporation, made a systematic study of the knocking characteristics of different hydrocarbons. More sophisticated engines had higher compression ratios – the fuel-air mixture was compressed more before it was ignited, but this also made it more likely that the mixture would pre-ignite, causing the engine to misfire – knocking – and lose power. At a particular point, a sparking plug fires a spark that ignites the mixture, and the resultant hot gases create the power that forces the piston down, driving the engine and powering the wheels. In a petrol engine, the gaseous mixture of air and hydrocarbon fuel is injected into the cylinder as the piston is on its downstroke, then gets compressed as the piston moves up. In the early days of the automobile, knocking or pre-ignition was a real problem. To understand that, you have to go back nearly a century.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
